  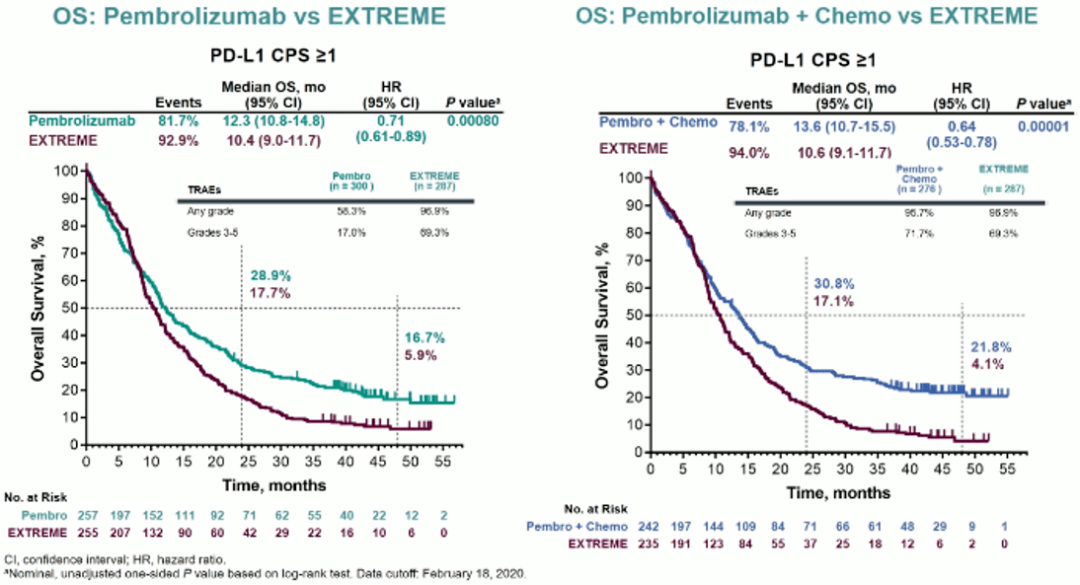
To further characterize the predictive value. Conclusions: Pembrolizumab monotherapy and pembrolizumab-chemotherapy extended OS while maintaining HRQoL, further supporting first-line use for R/M HNSCC. PURPOSE The phase III KEYNOTE-048 ( identifier: NCT02358031) trial of pembrolizumab in recurrent or metastatic (R/M) head and neck squamous cell carcinoma (HNSCC) included planned efficacy analyses in the total population and in participants with programmed death ligand-1 (PD-L1) combined positive score (CPS) 1 and CPS 20. TTD was similar between groups for EORTC QLQ-C30 GHS/QoL (pembrolizumab vs cetuximab-chemotherapy: HR, 1.38 95% CI, 0.95-2.00 pembrolizumab-chemotherapy vs cetuximab-chemotherapy: HR, 1.37 95% CI, 0.94-2.00), as was TTD in EORTC QLQ-H&N35 pain and swallowing scores. Median TTD in EORTC QLQ-C30 GHS/QoL and EORTC QLQ-H&N35 pain and swallowing scores was not reached over 51 weeks across groups, showing stable HRQoL. We present results from long-term follow-up of KEYNOTE-048, including analysis of progression-free survival on next-line therapy. At week 15, EORTC QLQ-C30 GHS/QoL scores remained stable no clinically meaningful between-group differences were observed (least squares mean difference, pembrolizumab vs cetuximab-chemotherapy, 0.24 95% CI, -3.34 to 3.82 pembrolizumab-chemotherapy vs cetuximab-chemotherapy, 0.40 95% CI, -3.46 to 4.26). On the basis of the results of the phase III KEYNOTE-048 study, pembrolizumab is now the standard of care for the first-line treatment of advanced head and neck squamous cell carcinoma (HNSCC). Results: Of 882 enrolled participants, 844 received ≥ 1 dose of study treatment and completed ≥ 1 HRQoL assessment adherence was ≥ 79% at week 15 across treatment groups. Secondary endpoints included mean change from baseline in EORTC QLQ-C30 global health status/quality of life (GHS/QoL) at week 15 and time to deterioration (TTD) in EORTC QLQ-C30 GHS/QoL and EORTC QLQ-H&N35 pain and swallowing. KEYNOTE-048 results show that pembrolizumab + chemotherapy was superior in overall survival versus EXTREME in the first line for patients with head and neck. Materials and methods: HRQoL was measured using the European Organisation for Research and Treatment of Cancer 30-question quality-of-life (EORTC QLQ-C30), the EORTC 35-question quality-of-life head and neck cancer-specific module (EORTC QLQ-H&N35), and the EuroQol 5-dimension 3-level instruments (EQ-5D-3L). The KEYNOTE-040 and -048 studies investigating pembrolizumab in advanced HNSCC both used the combined positive score (CPS), which is the number of PD-L1. Objectives: To assess health-related quality of life (HRQoL) with first-line pembrolizumab, pembrolizumab-chemotherapy, or cetuximab-chemotherapy in recurrent or metastatic head and neck squamous cell carcinoma (R/M HNSCC) in the phase 3 KEYNOTE-048 trial (NCT02358031). Pembrolizumab alone or with chemotherapy for recurrent or metastatic head and neck squamous cell carcinoma: health-related quality-of-life results from KEYNOTE-048 Please use this identifier to cite or link to this item: PURPOSEThe phase III KEYNOTE-048 ( identifier: NCT02358031) trial of pembrolizumab in recurrent or metastatic (R/M) head and neck squamous cell carcinoma (HNSCC) included planned.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
